The essential difference between ordinary steel and pure iron is
the amount of carbon in the former, which reduces the ductility but
increases the strength and the susceptibility to hardening when
rapidly cooled from elevated temperatures. On account of the various
micro-structures which may be obtained by different heat-treatments,
it is necessary to emphasise the fact that the following structures
are for "normal" steels, i.e. slowly cooled from 760-900°C depending
on the carbon contents.
The appearance of pure iron is illustrated in Fig. 1. It is only
pure in the sense that it contains no carbon, but contains very
small quantities of impurities such as phosphorus, silicon,
manganese, oxygen, nitrogen, dissolved in the solid metal. In other
words, the structure is typical of pure metals and solid solutions
in the annealed condition. It is built up of a number of crystals of
the same composition, given the name ferrite in metallography
(Brinell hardness 80).
The addition of carbon to the pure iron
results in a considerable difference in the structure (Fig. 2),
which now consists of two constituents, the white one being the
ferrite, and the dark parts representing the constituent containing
the carbon, the amount of which is therefore an index of the
quantity of carbon in the steel. Carbon is present as a compound of
iron and carbon (6-67 %) called cementite, having the chemical
formula Fe3
C. This cementite is hard
(Brinell hardness 600 +), brittle and brilliantly white.
|

x200
Figure 1.
Armco iron: ferrite
|

x200
Figure 2.
0,4% carbon steel. Ferrite +
pearlite |
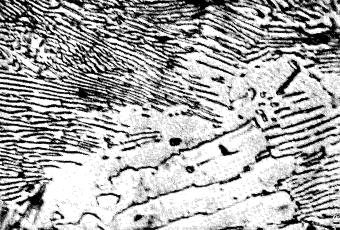
On examination the dark parts will be seen
to consist of two components occurring as wavy or parallel plates
alternately dark and light (Fig. 3). These two phases are ferrite
and cementite which form a eutectic mixture, containing 0,87% carbon
and known as pearlite. The appearance of this pearlite depends
largely upon the objective employed in the examination and also on
the rate of cooling from the elevated temperature.
 |
|
Figure 3.
0,87% carbon
steel |
Allotropy of iron
Certain substances can exist in two or more
crystalline forms; for example charcoal, graphite and diamonds are
allotropic modifications of carbon. Allotropy is characterized by a
change in atomic structure which occurs at a definite transformation
temperature.
Four changes occur in iron, which give rise
to forms known as alpha, beta, gamma and delta. Of these, a, b and d forms have the same atomic structure (body
centred cubic) while g
-iron has a face
centred cubic structure. Iron can, therefore, be considered to have
two allotropic modifications.
The A2 change at 769°C, at which the a-iron loses its magnetism, can be ignored from a
heat-treatment point of view. These changes in structure are
accompanied by thermal changes, together with discontinuities in
other physical properties such as electrical, thermo-electric
potential, magnetic, expansion and tenacity. The A3
change from a b.c.c. to an f.c.c. atomic structure at 937°C is
accompanied by a marked contraction while the reverse occurs at
1400°C. These changes in structure are accompanied by
recrystallisation, followed by grain growth.
Critical points
The addition of carbon to iron, however, produces another
change at 695°C, known as A1 and associated with the formation of
pearlite. These structural changes, which occur during cooling, give
rise to evolutions of heat, which cause arrests on a cooling curve.
The temperatures of these arrests are known as critical points or
"A" points. These arrests occur at slightly higher temperatures on
heating, as compared with cooling, and this lag effect, increased by
rapid cooling, is known as thermal hysteresis.
To differentiate between the arrests
obtained during heating and cooling, the letters c and r
respectively are added to the symbol A (from chauffage and
refroidissement). In a steel containing about 0,8-0,9% carbon the
evolution of the heat at Ar1
is sufficient to
cause the material to become visibly hotter and the phenomenon is
called "recalescence".
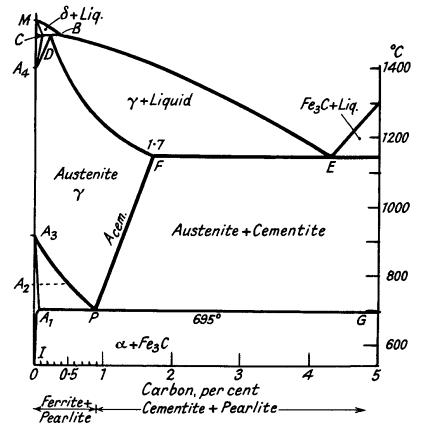
Iron-cementite equilibrium diagram
The addition of carbon to iron not only
gives rise to the A1 point but also influences the
critical points in pure iron. The A4 point is raised; and
the A3 point lowered until it coincides with
A1. The a, b and d modifications,
which may be called ferrite, have only slight solubility for carbon,
but up to 1,7% of carbon dissolves in y-iron to form a solid
solution called Austenite. These effects are summarised in the
iron-Fe3
C equilibrium diagram (Fig.
4), which is of much importance in the study of
steels.
The iron-iron carbide system is not in true equilibrium, the
stable system is iron-graphite, but special conditions are necessary
to nucleate graphite. Will be seen that the complicated
Fe-Fe3C diagram can be divided into several simple
diagrams:
Peritectic transformation CDB - d-iron
transforms to austenite. Eutectic at E - austenite and cementite.
Solid solution D to F - primary dendrites of austenite form.
Eutectic point at P - formation of pearlite.
|
|
|
Figure 4.
Iron-cementite equilibrium
diagram
|
The ferrite solubility line,
A3P, denotes the commencement of precipitation of ferrite
from austenite. The cementite solubility line, FP,
indicates the primary deposition of cementite from austenite.
The pearlite line, A1PG, indicates the formation
of the eutectic at a constant temperature. Let us consider the
freezing of alloys of various carbon contents.
0,3% carbon
Dendrites of d-iron form, the
composition of which is represented eventually by C (0,07 %), and
the liquid, enriched in carbon, by B. The solid crystals then react
with the liquid to form austenite of composition D. Diffusion of
carbon occurs as the solid alloy cools to line A3P. Here
a-ferrite commences to be ejected from the
austenite, consequently the remaining solid solution is enriched in
carbon, until point P is reached at which cementite can be also
precipitated.
The alternate formation of ferrite and cementite at 695°C gives
rise to pearlite. The structure finally consists of masses of
pearlite embedded in the ferrite.
0,6% carbon
When line BE is reached dendrites of austenite form, and finally
the alloy completely freezes as a cored solid solution, which, on
cooling through the critical range (750-695°C), decomposes into
ferrite and pearlite.
1,4% carbon
Again, the alloy solidifies as a cored solid solution, but on
reaching line FP, cementite starts to be ejected and the residual
alloy becomes increasingly poorer in carbon until point P is
reached, when both cementite and ferrite form in juxtaposition. The
structure now consists of free cementite and pearlite.
List of Articles - Knowledge Base